 
J-codes standardize the submission and payment of insurance claims across Medicare, Medicare Advantage, Medicaid and commercial insurance plans. In July, CMS awarded a product-specific permanent J-code for OMIDRIA, which will become effective October 1, 2019. Recent developments regarding OMIDRIA include the following: Each of these unique and cutting-edge programs is focused on significantly improving – or saving – patients’ lives.” As OMIDRIA ramps toward fully funding our pipeline, our assets increasingly declare their value – narsoplimab is moving toward anticipated approval and launch, OMS527 for addiction has successfully completed the Phase 1 clinical trial, OMS906 targeting the alternative complement pathway and our follow-on MASP-2 inhibitors are slated to enter the clinic beginning next year, and GPR174 inhibition appears to play a key role in cancer immunotherapy. “Indications are that sales will continue to grow, helped in part by our new permanent J-code, broadening Med Advantage and commercial payer reimbursement, and additional strong clinical data that we believe places OMIDRIA squarely within CMS’ criteria for separate payment. Demopulos, M.D., Omeros’ chairman and chief executive officer.  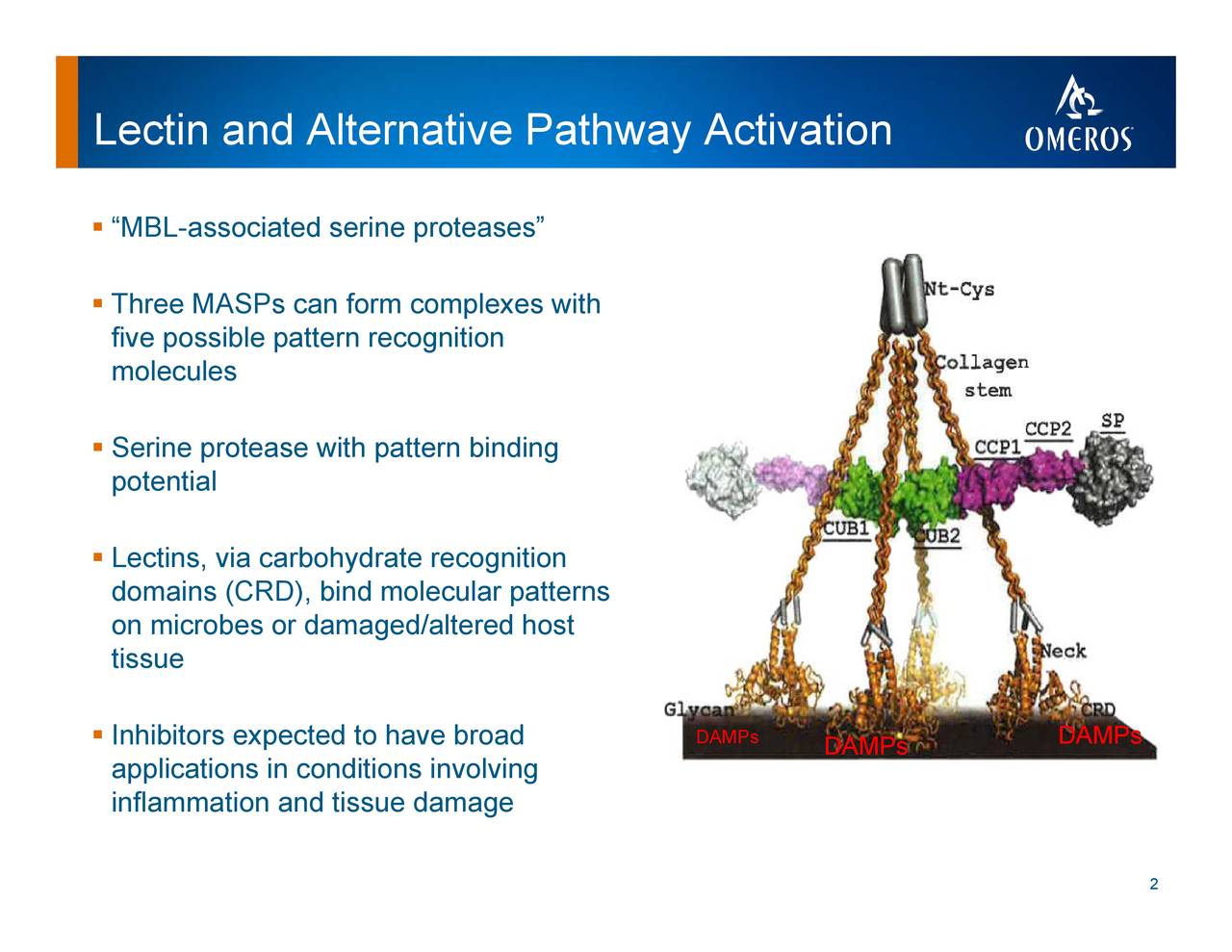
“OMIDRIA revenues continue to set new quarterly records as our customer base continues to broaden throughout all channels and our per-account capture of cataract procedures grows,” said Gregory A. The J-code will become effective October 1, 2019. Received a product-specific permanent J-code for OMIDRIA. Reached agreement with FDA on the primary endpoint criteria for the pivotal trial to support the biologics license application (BLA) for narsoplimab to treat hematopoietic stem cell transplant-associated thrombotic microangiopathy (HSCT-TMA).Īs part of a successful pre-BLA meeting directed to chemistry, manufacturing and controls (CMC), the company discussed with FDA the CMC requirements for the narsoplimab BLA for HSCT-TMA and is confident in its ability to meet them.Įxecuted a long-term commercial manufacturing agreement with Lonza in preparation for market launch of narsoplimab. Borrowing availability is based on eligible accounts receivable, subject to applicable reserves. In August 2019, the company entered into a $50-million revolving line of credit facility with Silicon Valley Bank. This compares to a net loss of $24.3 million, or $0.50 per share, in 1Q 2019.Īt June 30, 2019, Omeros had cash, cash equivalents and short-term investments available for operations of $31.8 million and an accounts receivable balance of $28.5 million. Net loss in 2Q 2019 was $14.5 million, or $0.29 per share, which included non-cash expenses of $6.3 million, or $0.13 per share. The increase of $5.0 million, or 23 percent, over the prior quarter reflects both an expanded number of purchasing accounts and deeper penetration across Ambulatory Surgery Centers (ASCs), hospitals and the Veterans Administration and other government systems. This compares to $21.8 million in 1Q 2019. OMIDRIA ® revenues for 2Q 2019 were a record high at $26.8 million. 
Omeros Corporation (Nasdaq: OMER), a commercial-stage biopharmaceutical company committed to discovering, developing and commercializing small-molecule and protein therapeutics for large-market as well as orphan indications targeting inflammation, complement-mediated diseases, disorders of the central nervous system and immune-related diseases, including cancers, today announced recent highlights and developments as well as financial results for the second quarter ended June 30, 2019. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
